 Problems such as poor transit or constipation are common, and can produce significant misery for the individual compromised in this manner. Butyrate is a short chain fatty acid, manufactured in the gut by the anaerobic fermentation of dietary fibres by resident microbiota. It is proposed that apart from its already well understood properties that it has another remarkable effect – the ability to increase the neuronal concentration of the Enteric Nervous System.[1]
Problems such as poor transit or constipation are common, and can produce significant misery for the individual compromised in this manner. Butyrate is a short chain fatty acid, manufactured in the gut by the anaerobic fermentation of dietary fibres by resident microbiota. It is proposed that apart from its already well understood properties that it has another remarkable effect – the ability to increase the neuronal concentration of the Enteric Nervous System.[1]
Butyrate-generating foods and supplements might become an effective and simple option to prevent or treat functional gut disorders via modulation of enteric neuroplasticity.
The Enteric Nervous System (ENS) participates in the regulation of gut immune and inflammatory processes. In addition, the ENS is responsible for a wide range of reparative, maintenance, and adaptive properties, activated in response to different physiological (e.g., development and aging) or noxious stimuli (e.g., inflammation, infectious agents, and toxins). The ENS, like the brain is subject to pressures that result in altered neural plasticity, providing a therapeutic opportunity to correct problems linked to these tissues. Nutritional components have been shown to alter cognition and it is this feature that has attracted the idea that a similar mechanism may be available to correct aberrations in the ENS.[2]
The major SCFA in humans are butyrate, propionate, and acetate, where butyrate is the major energy source for colonocytes, propionate is destined for uptake by the liver, and acetate enters the peripheral circulation to be metabolised by peripheral tissues. A novel understanding of the role of butyrate on colonic transit is being uncovered.[3]
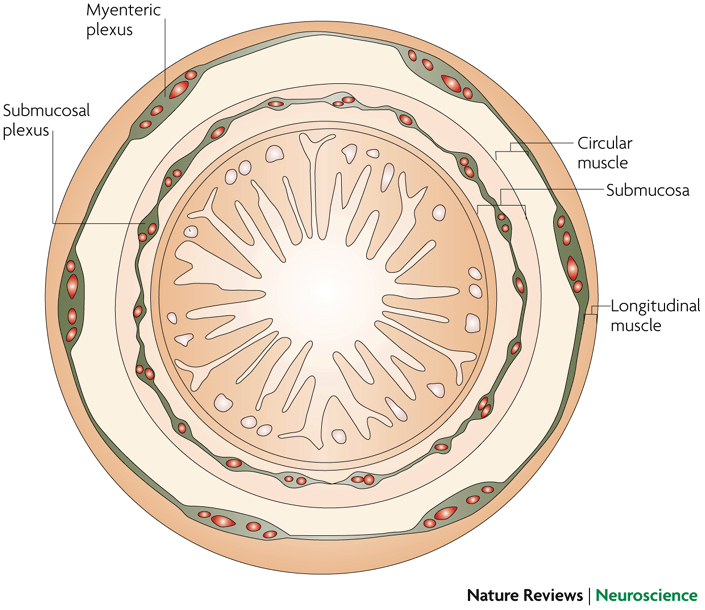
The idea that long-term exposure to dietary factors, such as the SCFA butyrate, may impact positively on colonic transit by specialised phenotypic changes of myenteric neurons (found between the longitudinal and circular muscle layers of the muscularis externa), requires understanding of two important and intensively investigated multidisciplinary research areas:
- The contribution that prebiotics (i.e., fermentable substrates), probiotics (i.e., bacteria that can establish temporary residency in the gut lumen), and synbiotics (i.e., mixtures of prebiotics and probiotics) may provide to the therapeutic management of functional GI disorders acting as sources of nutritional factors.[4]
- The role that the bacterial flora resident in the gut lumen, collectively known as “gut microbiota,” play in the digestion of dietary components and maintenance of colonic health.[5]
The intake of fibre and especially soluble fibres have been shown to have powerful immune related benefits in the animal model, as described in this previous post. Recommending dietary supplementation with fibres, such as oligofructose, oat bran, and apple pectin, is known to promote a significant production of SCFA upon fermentation by saccharolytic bacteria (Capable of hydrolysing or otherwise breaking down a sugar molecule) in the gut lumen.
Novel View
In the long term, the beneficial effects of these fibres, with particular regard for the shortening of colonic transit time, might not depend, as commonly believed, solely on the mechanical stress of faecal bulk on the gut wall, but rather result from sophisticated mechanisms related to targeted rearrangements of gene expression and phenotypic changes in myenteric motor neurons.
Scientists and clinicians are increasingly appreciating that the human gut microbiota represents an “extended genome,” comprising millions of microbial genes (i.e., the gut microbiome). This organ within an organ through a complex symbiosis, influence the patients metabolism, physiology, and gene expression. Advances in microbiologic analysis are beginning to implicate the gut microbiome in the pathogenesis of different intestinal diseases and other complicated systemic health conditions.
Conclusion
Overall, the scientists have uncovered solid evidence that a dietary factor, such as butyrate, can increase the proportion of cholinergic neurons in myenteric ganglia in rats, thereby enhancing colonic propulsive motility. The implications are that subject to increased studies that this feature can provide the basis for the development of novel therapeutic strategies in the field of GI disorders associated with delayed colonic transit.
Comment
The exogenous supplementation with a prepared form of Butyrate has long been part of the gastrointestinal repair programme and may represent a novel strategy for improving long term mucosal nervous system function. In conjunction with suitable soluble fibres and probiotics the combination has the potential to offer a gene based treatment using food extracts for resolution of a common and uncomfortable problem. Poor dietary intake of suitable fibres, especially in women may contribute to a reduction in SCFA’s leading to a diminished programming of the mucosal ENS. Consider when managing patients with this condition that butyrate and fibre such as rye bread plus LGG may provide short and long term resolution and gene expression modification.[6]
References
[1] Soret R, Chevalier J, De Coppet P, Poupeau G, Derkinderen P, Segain JP, & Neunlist M (2010). Short-chain fatty acids regulate the enteric neurons and control gastrointestinal motility in rats. Gastroenterology, 138 (5), 1772-82 PMID: 20152836
[2] Gómez-Pinilla, F. (2008). Brain foods: the effects of nutrients on brain function Nature Reviews Neuroscience, 9 (7), 568-578 DOI: 10.1038/nrn2421
[3] de Giorgio R, & Blandizzi C (2010). Targeting enteric neuroplasticity: diet and bugs as new key factors. Gastroenterology, 138 (5), 1663-6 PMID: 20332039
[4] Sherman PM, Ossa JC, Johnson-Henry K. Unravelling mechanisms of action of probiotics. Nutr Clin Pract. 2009 Feb-Mar;24(1):10-4. View Abstract
[5] Wong JM, de Souza R, Kendall CW, et al. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol. 2006;40:235–243. View Abstract
[6] Hongisto SM, Paajanen L, Saxelin M, Korpela R. A combination of fibre-rich rye bread and yoghurt containing Lactobacillus GG improves bowel function in women with self-reported constipation.Eur J Clin Nutr. 2006 Mar;60(3):319-24. View Abstract






3 Comments. Leave new
How we can purchase butyrate enema
Dear Dr Mohamed.
It is quite possible to purchase butyrate via various supplement manufacturers in capsule form. This may also be added to enemas. However, my experience is that Butyric acid bound to magnesium and taken orally works just as well and has a higher compliance.
How can I purchase Butyric Acid